If an atom could change its number of protons then it would become a different type of atom. A better way of saying it is for an atom to be a gold atom it must have 79 protons, no more and no less. All atoms that have 6 protons are carbon atoms. For example, all atoms that have 8 protons in the nucleus are oxygen atoms. 
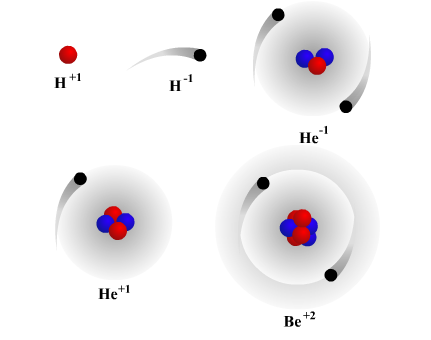
The primary sub-atomic particle is the proton, because the number of protons inside an atom determines which type of atom it is. The Number of Protons Determines the Type of Atom You can see that this true scale model shows that there is a tremendous amount of space that comprises atoms and therefore atoms are mainly made of space. If we could make a true-to-scale model of an atom we would see that the nucleus is very small, let’s say the size of a baseball, and the electrons are like the size of raisins spinning around the baseball from a very far distance of around 5 football fields away, or 1,500 ft. We are familiar with electrons in the sense that electrons flow through wires as electricity.Įlectrons orbit the nucleus at different distances from the nucleus, with the more energetic electrons orbiting further away. The electron holds a negative electric charge. The electrons orbit the atom in a large region of space surrounding the nucleus. The 3rd type of sub-atomic particle is called the electron and it is much smaller than the proton or the neutron. Neutrons are also arranged in the nucleus and these particles have no electrical charge and hence are neutral. The second type of particle in the nucleus is called the neutron. The center of the atom is called the nucleus. The proton is a particle that carries a positive electric charge and is arranged in the center of the atom. Part 1 - Atomic Struction - Protons, Neutrons and Electrons Inside an atom are three types of sub-atomic particles: protons, neutrons and electrons. If an atom is divided then it can no longer be recognized as Students the entire universe is made up of extremely small particles of matter called atoms. Panico’s Geoscience Course - Odyssey Charter School - Sept. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
